
UTMB Neonatology Manual
Babies with Special Challenges
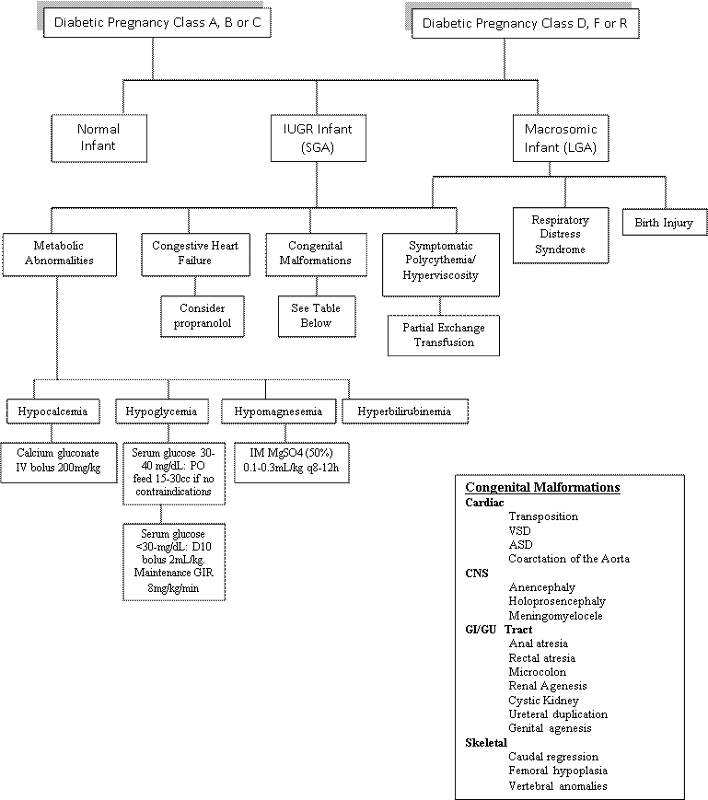
The Infant of the Diabetic Mother

Significant advances have been made in reducing perinatal mortality for the infant born to the mother with diabetes mellitus.
However, the infant of the diabetic mother (IDM) is likely to manifest a variety of problems, all of which require anticipation, recognition, and appropriate therapy.
Knowledge of the mother's diabetes, prior medical and pregnancy history, and complications during this pregnancy permits anticipation of many of the problems likely to be present in the postnatal period.
White's Classificaton permits assessment of severity of maternal diabetes. Classes B-H require medication.
Class A:
Chemical diabetes: positive glucose tolerance tests prior to or during pregnancy.
Prediabetes: history of large babies more than 4 kg or unexplained stillbirths after 28 weeks. Any age of onset or duration.
Class B: Onset after 20 years of age; duration less than 10 yrs.
Class C:
C1: Onset at 10-19 years of age.
C2: Duration 10-19 years.
Class D:
D1: Onset before 10 years of age.
D2: Duration 10 years.
D3: Calcification of vessels of the leg (macrovascular disease).
D4: Benign retinopathy (microvascular disease).
D5: Hypertension
Class E: Same as D, but with calcification of pelvic vessels.
Class F: Nephropathy
Class G: Many reproductive failures.
Class H: Diabetic cardiomyopathy
Class R: Malignant retinopathy
Class RF: Both nephropathy and retinopathy
Pedersen's prognostic signs of pregnancy permit more accurate predictions of poor outcome. These are: clinical pyelonephritis, precoma or severe acidosis, toxemia and neglectors (women who are uncooperative with the treatment plan.)
Diabetic women have normal fertility. In the first trimester there is an increased rate of spontaneous abortions. The incidence of these early losses varies with the diabetic classification:
Class A, 5-10% (not significantly different from the general population); Class B, 10%; Class C, 24%; Class D, 30%; and Class E, 74%.
Good metabolic control of maternal diabetes improves the perinatal outcome.
Timing of delivery is individualized in each case and requires decision based on the risks of in utero fetal distress and stillbirth compared to the problems of prematurity.
Types of tests:
Once the baby has been delivered, a careful assessment made on the basis of the Apgar score should indicate the need for any resuscitative efforts. In the delivery room, a screening physical examination for major congenital anomalies should be performed.
In the nursery, a more complete physical examination with special attention to the heart, kidneys, and extremities is performed. Close observation for potential problems should begin.
Intrauterine Growth Restriction (IUGR) Not Due to Intrauterine Viral Infection or Chromosomal Abnormality
Each newborn infant should have his weight, length, and head circumference plotted by gestational age on the intrauterine growth curve. An infant whose weight is below the 10th percentile for his gestational age (see chart) may have intrauterine growth restriction and is at risk for hypoglycemia and polycythemia. These babies should be managed as follows:
Infants of more than 42 weeks gestation are considered to be post-term. Those who show the typical physical findings but are less than 42 weeks gestation should be regarded as post-mature.
Management should be the same as for IUGR with regard to Hct and blood glucose.
Post-mature infants are likely to have meconium in the amniotic fluid and are at risk for meconium aspiration (See Section on Meconium Aspiration).
Section references: http://www.emedicine.com/med/topic3247.htm http://www.Netsciv.org.au/nets/handbook/index.cfm?doc-ie=821
Micropremies
Infants of <1000 g birthweight are defined as extremely low birthweight (ELBW), and are sometimes referred to as micropremies. These guidelines address the exquisite fragility of these infants with regard to skin integrity, fluid/electrolyte status, ability to regulate stimuli and sheer smallness of body size. Reduction of noxious stimuli is particularly important in the prevention of intraventricular hemorrhage. Remember to check with each attending for approval of micropremie admission orders for each infant.
Most of the guidelines below are included in the EMR order sets. ( * = not in order set)
1. Weigh on admission, on the fifth day of life (4 days old), and then daily.
2. Nursing physical assessments should be done every 12 h and with obvious changes in status. Vital signs are recorded from the monitors as ordered (every 1-4 hours).
3. Infants who are on ventilators should receive endotracheal suctioning 1-2 hours after surfactant administration and then only as needed.
(In the first several days, fluid orders should be assessed q 6-8 hrs.)
The micropremie is at particular risk for hypernatremia, hyperkalemia, hypocalcemia, hypo- or hyperglycemia and hyperbilirubinemia. The need for laboratory monitoring must be balanced
with the need to minimize blood transfusion (which worsens hyperbilirubinemia).
1. 0.45% NaCl (1/2 normal saline) should be used for all intravascular flushes.
2. Continuous calcium infusion from birth (~200 mg/kg/day) will usually prevent the early hypocalcemia, which typically occurs at 24-48 hours.
3. Saran wrap or a protective shield should be placed over infants on open warmers.
4. Arterial blood gases with electrolytes, glucose, and hematocrit (deluxe gas) every 8 hours x 24h
5. Basic metabolic panel, magnesium and phosphorus and bilirubin at 12 hours, then every day for three days.
*While on TPN, obtain BMPs once-twice weekly.
If on PN > 4 weeks, obtain liver function tests (AST, ALT, GGT, bili, protein, albumin) every other week.
6. Prophylactic phototherapy beginning after stabilization at admission.
7. Insulin infusion may be required to control excessive hyperglycemia.
1. Minimize tape use, particularly in constructing the umbilical vessel catheter bridge, and use baby tape or duoderm.
2.The temp probe cover is cut in quarters and only one quarter is used to secure the temperature probe on the baby.
3. For cardiac monitoring, micro arbo leads or limb leads are used.
4. *In most situations, the UAC or UVC can be used for transfusion of blood and infusion of medications.
5. Protect skin by application of Aquaphor ointment after birth, and continue for 7 days.
1. *The catheters should be size 3.5 in infants less than 1500g.
2. *The tip of the UAC should be in the high position (T6-T9) to facilitate monitoring of correct placement and to decrease risk of peripheral embolic phenomenon.
1. Admission Solution should be started at birth (contains protein, glucose, calcium).
2. If the baby is born before 5 PM, intralipid infusion at 0.5 g/kg/day should be started on the day of birth.
3. TPN should be started on the first or second day of life.
4. Consider using UAC fluid containing acetate (#3), if available. Sodium acetate is among the drugs affected by intermittent nationwide shortages of supply.
5. The baby will usually need help with meconium evacuation. In a step-wide fashion, interventions include:
- glycerin suppository
- normal saline enema
- serial n-acetylcysteine (Mucomyst) enemas and oral Mucomyst
(* = not in UTMB EMR order sets)
Multiple Gestations
The incidence of monozygotic (i.e. identical) twins (MZ) remains constant worldwide and is often quoted at 3-5 per 1000. However, the incidence of dizygotic twins (DZ) varies widely in different populations and under different circumstances (4-50 per 1000). These figures may be underestimations, because they do not account for the much higher prenatal mortality of twins (as abortions, fetus papyraceus, vanishing twin phenomena). When the twinning rate of a population is known, the frequencies of other types of multiple gestations can be roughly calculated by employing Mellein's hypothesis: when the frequency of twinning is n, that of triplets is n2, of quadruplets n3, etc.
Dizygotic twins account for 2/3 of twins and refers to the fertilization of 2 ova. The resulting blastocysts generate separate amnions, chorions and placentas. The placenta will be dichorionic, diamniotic separate if implantation sites for apart or dichorionic, diamniotic fused if the implantation sites are proximate. The current, incomplete understanding of dyzygotic twinning is that it results from elevated maternal serum gonadotropin levels, which in turn is influenced by maternal age, parity, nutrition and genotype.
Monozygotic twins account for 1/3 of twins and result from the splitting of a single fertilized ovum sometime during the 1st 2 to 15-17 days gestation. Because MZ twins occur more frequently with advancing maternal age, are associated with congenital malformations and can be induced by teratogens, it has been hypothesized that MZ twins result from a teratogenic event.
If the division of the ovum occurs early (blastomere stage) the placenta is dichorionic,diamniotic. If splitting occurs between 4-7 days, when duplication of the inner core mass and differentiation of the trophoblast to form the chorion occurs, but before formation of the amnion, the placenta will be monochorionic, diamniotic (20-30% of MZ twins). With duplication of the embryonic rudiment of the embryonal disc at day 15 or 16, after the amnion has formed, the placenta will be monochorionic, monoamniotic (1-2% of MZ).
Later or incomplete splitting of the embryonal disc leads to the phenomena of conjoined twins.
Multiple gestation results in increased mortality and morbidity in both mother and babies.
1. Increased rates of prolonged hospitalization and surgical deliveries. Decisions concerning delivery are based on many factors, not the least of which is fetal position:
Vertex vertex: vaginal delivery is appropriate.
Breech vertex: cesarean section, because of increased chance of entrapment of the aftercoming twin and cord prolapse.
Vertex breech: controversial.
2. The rate of spontaneous abortion is twice that of singleton pregnancies. Loss of one twin occurs in 5% of twin pregnancies.
3. Increased incidence of hyperemesis gravidarium.
4. Exaggerated anemia of pregnancy.
5. Polyhydramnios which can lead to premature and/or prolonged ROM.
6. Preterm labor - 50% of twins gestations deliver before 37 weeks (12X higher than that in singletons).
7. Increased incidence of pre-eclampsia (5X that of singletons).
8. Increased risk of maternal hemorrhage, primarily from uterine atony due to over-distention of uterus, but also from the increased incidence of placenta previa and vasa previa.
Malpresentation, cord prolapse, cord entanglement, and fetal distress are more common. Locking or collusion of twins can occur but is rare.
1. Perinatal mortality in a twin pregnancy is about 5X higher than a singleton pregnancy with a slightly higher risk for the 2nd twin. The increased risk of death in twins persists during the 1st year of life.
2. Increased preterm deliveries - 10% of preterm deliveries are twin gestations. They also account for 25% of perinatal deaths in preterm deliveries, the majority occurring with gestation < 32 weeks and birthweight < 1500g.
3. Incidence of low birth weight (< 2500 g) is greater in twin deliveries (> 50%). Low birth weight results both from prematurity as well as from IUGR.
4. The intrauterine growth of the twin fetus follows the pattern of growth of singletons until 30-34 weeks gestation, thereafter the mean birth weight for twins deviates from the curve for singletons. Peak of mean birth weight is thus reached at 37-38 weeks rather than 41-42 weeks as seen in singletons. The decrement in intrauterine growth is evident in most major organs (heart, lungs, kidneys, liver, spleen and brain). Head circumference and body length are better maintained until later in pregnancy. MZ twins tend to be lighter than DZ twins and 2nd born twins are reported to weigh slightly less than the 1st born twin.
5. Twins are more prone to birth asphyxia, and within twin pairs the 2nd born twin is more susceptible.
6. The second twin is more likely to develop RDS.
7. The incidence of neurodevelopmental disorders is increased in twins as compared to singletons. Cerebral palsy is more frequent in twins (5-10%), and the most common form of palsy is spastic diplegia. Twins also have a higher incidence of seizures. Twins continue to exhibit significant delays in growth and development postnatally that can last into the preteens.
8. Retinopathy of prematurity is increased in infants of multiple births compares with singletons.
9. Necrotizing enterocolitis is 3X higher in twins and triplets.
10. The rate of early onset GBS infection in LBW infants is nearly 5X higher in twins than singletons.
11. Congenital abnormalities are also increased in multiple gestations. Congenital malformations may be related to:
a. Crowding/limited intrauterine space leading to improper growth and positioning and can result in defects such as talipes and congenital hip dislocation
b. Defects resulting from the specific pathology/teratogenicity of the twinning process including conjoined twins, CNS abnormalities such as neural tube defects, CVS anomalies such as single umbilical artery, and GI anomalies.
c. Malformations resulting from vascular interchange.
Vascular anastomoses are found almost exclusively in monochorionic placentas. An incidence of 85% has been reported.
When a cotyledon is supplied by an artery from one twin and drained by a vein from the other, the potential exists for transfer of significant quantities of blood from the twin on the arterial side (donor) to the twin on the venous side (recipient) of the connection. The effect of feto-fetal transfusions on each of the twins probably depends on the period over which the transfer of blood occurs. Clinically, TTTS can be suspected when twins exhibit a Hb difference of >5g/dl and/or birthweight difference of > 20%. Ultrasonographic evidence may be seen even if there is no difference in hematocrits.
In acute cases, one twin is plethoric and the other pale. In chronic cases, significant growth retardation of the donor twin may be complicated by oligohydramnios with concominant polyhydramnios of recipient twin (due to the combined effects of hypervolemia, hypertension and increased urine output). In severe cases, hydrops fetalis has been described.
The most devastating effect of vascular connections in a monochorionic placenta is the formation of an acardiac or amorphous fetus (1% of all MZ twins). This occurs in association with a large A-A and V-V anastomosis between the anomalous fetus and its usually normal co-twin. Early in gestation the pressure in the artery of the normal twin exceeds that in the artery of the other. Blood enters the recipient through an umbilical artery and leaves through the umbilical vein to drain into a large vein-vein anastomosis. The circulation of the recipient twin is thus reversed. (Remember that oxygenated blood normally flows into the fetus from an umbilical vein and desaturated blood exits via the umbilical arteries.)
Low perfusion pressure, desaturation and circulatory reversal in the recipient leads to multiple, severe structural defects producing an acardiac or amorphous fetus. The cephalic pole is more severely affected because it is the region most distal to retrograde perfusion.The closer and better perfused lower part of the body is relatively spared. As the initially normal donor twin must perfuse both fetal circulations, it may develop cardiac hypertrophy, congestive cardiac failure or hydrops fetalis.
In addition to the transfer of blood, vascular anastomoses can serve as channels for the passage of thromboplastic substances. Following the intrauterine demise of a co-twin, the transfer of thromboplastic material to a living recipient can initiate widespread intravascular coagulation with resultant thrombosis, infarction and necrosis in the organs of that recipient. This sequence of events may produce intrauterine death, renal cortical necrosis, encephalomalacia and disruptive structural defects such as intestinal atresia, aplasia cutis or limb amputation.
1. Maintain adequate ventilation, blood pressure, glucose and electrolyte levels.
2. Maintain optimal thermal environment.
3. Assess newborn carefully for anomalies, IUGR, anemia and coagulopathy.
4. In cases of acute fetofetal transfusions, with evidence of volume overload in recipient and shock in donor, exchange transfusion may be required. Estimated blood volume (EBV) is 100 ml/kg for preterm babies and 85 ml/kg for term babies.
a) Partial exchange transfusion using FFP for recipient.
Volume of exchange (mLs) = EBV (mLs) x desired Hct change
Starting Hct
b) Rapid volume expansion with crystalloid or colloid solution followed by PRBC transfusion for the donor.
Volume of Exchange = 16 g/dL(hemoglobin of PRBC) - (initial Hb - desired Hb)
2
5. In cases of chronic fetofetal transfusion:
a) Partial exchange transfusion using PRBC if donor is anemic and symptomatic.
b) If donor has significant growth restriction and is unstable, withhold enteral feeds and start continuous infusion of dextrose to initially provide 5 mg/kg/min of glucose. This can subsequently be adjusted based on blood glucose measurements.
6. In both acute and chronic fetofetal transfusions, monitor the recipient twin for hyperbilirubinemia.
7. If there is evidence of cardiac decompensation, fluid restriction, use of diuretics and pressor agents may be necessary. Estimation of intravascular volume by CVP monitoring may also be helpful.