
In his
April 29 Blog, NIH Director, Dr. Francis Collins, outlined his belief that biomedical technology has a vital role to play in trying to contain the global coronavirus disease 2019 (COVID-19) pandemic. As an answer, NIH launched the Rapid Acceleration of Diagnostics (RADx) Initiative.
This new initiative is aimed at speeding innovation, development and commercialization of COVID-19 testing technologies. It will infuse funding into early innovative technologies to speed development of rapid and widely accessible COVID-19 testing. At the same time, NIH will seek opportunities to move more advanced diagnostic technologies swiftly through the development pipeline toward commercialization and broad availability. NIH will work closely with the U.S. Food and Drug Administration, the Centers for Disease Control and Prevention and the Biomedical Advanced Research and Development Authority (BARDA) to advance these goals.
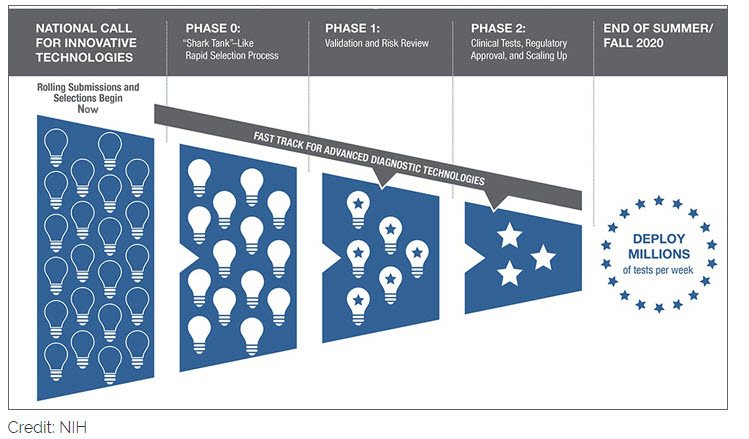
As part of this initiative, NIH is urging all scientists and inventors with a rapid testing technology to compete in a national COVID-19 testing challenge for a share of up to $500 million over all phases of development. The technologies will be put through a highly competitive, rapid three-phase selection process to identify the best candidates for at-home or point-of-care tests for COVID-19. Finalists will be matched with technical, business and manufacturing experts to increase the odds of success. If certain selected technologies are already relatively far along in development, they can be put on a separate track and be immediately advanced to the appropriate step in the commercialization process. The goal is to make millions of accurate and easy-to-use tests per week available to all Americans by the end of summer 2020, and even more in time for the flu season.
RADx will expand the Point-of-Care Technologies Research Network(link is external) (POCTRN) established several years ago by NIBIB. The network will use a flexible, rapid process to infuse funding and enhance technology designs at key stages of development, with expertise from technology innovators, entrepreneurs and business leaders across the country. POCTRN supports hundreds of investigators from multiple universities and businesses through five technology hubs:
- Emory University/Georgia Institute of Technology, Atlanta
- Johns Hopkins University, Baltimore
- Northwestern University, Evanston, Illinois
- University of Massachusetts Medical School, Worcester
- Consortia for Improving Medicine with Innovation & Technology (CIMIT) at Harvard Medical School/Massachusetts General Hospital, Boston
RADx is focused on diagnostic testing, but NIH is also intensely engaged in developing safe, effective therapies and vaccines for COVID-19. One innovative effort, called
Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV), is a public-private partnership that aims to speed the development of ways to treat and prevent this disease that’s caused so much suffering and death around the globe.
More information can be found in the following links:
NIH mobilizes national innovation initiative for COVID-19 diagnostics
NIH news release, April 29, 2020
Point-of-Care Technologies Research Network
(National Institute of Biomedical Imaging and Biotechnology/NIH)
NIH to launch public-private partnership to speed COVID-19 vaccine and treatment options
NIH news release, April 17, 2020