Embryology
Snigdha Bhatia MBBS, Saef Munir, Ashraf Aly, MD
Early Development
- Development of the heart starts during the 3rd week of gestation and is completed by the 9th week.
- A single tube is formed with a venous and an arterial end.
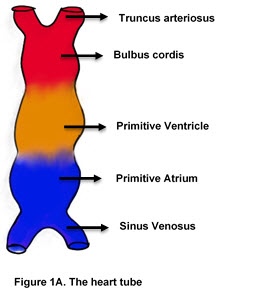
- The heart tube has five distinct areas (going from up to down- Truncus arteriosus, Bulbus cordis, Primitive Ventricle, Primitive Atrium and the Sinus Venosus) (Figure 1A).
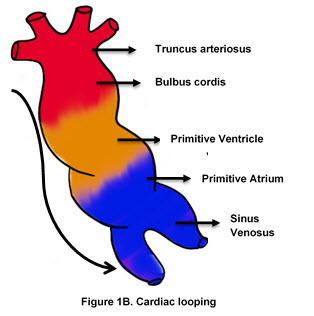
- Cardiac looping begins around the 22nd day of intra-uterine life. It results from elongation of the cardiac tube while both ends of the tube are fixed.
- Left-right polarity is established when the primitive ventricle is bent into a loop that moves anteriorly and to the right. At the same time, the caudal portion of the tube bends dorsally, cephalically, and to the left. (Figure 1B)
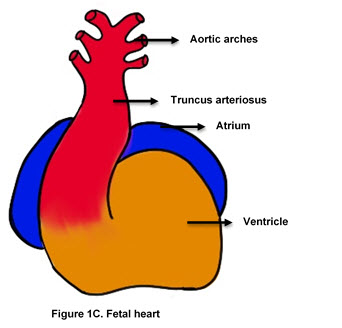
- By day 28, heart consists of the common atrium, common ventricle, and common outflow tract. (Figure 1C)
- During this time, blood is pumped from the distal end through the sinus venosus upwards to the truncus arteriosus.
- Development of the four chambered heart with two outflow tracts involves simultaneous formation of three septa that separate atria, truncus arteriosus, and ventricles.
|
|
|
|
|
Embryonic Heart Structure |
Final Heart Structure |
|---|---|
|
Truncus arteriosus |
Ascending aorta and pulmonary trunk |
|
Bulbus cordis |
Smooth portions of right and left ventricle |
|
Primitive ventricle |
Trabeculated right and left ventricle |
|
Primitive atrium |
Anterior (trabeculated) portions of right and left atria |
|
Sinus venosus |
Posterior portion of right atrium and coronary sinus |
|
Endocardial cushions |
Aortic, pulmonary, tricuspid, mitral valves |
Atrial Septation
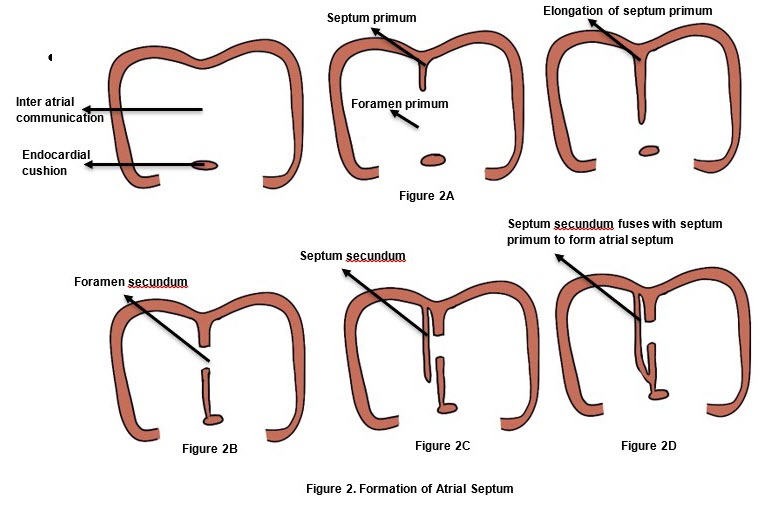
- Primitive atrium is a single cavity. The primary atrial septum (septum primum) appears as a thin walled sagittal fold in the roof of the common atrium and grows down towards the endocardial cushions.
- The opening between the septum and the endocardial cushions is called foramen primum. (Figure 2A). Tissue resorption at the superior end of the septum primum results in formation of foramen secundum (Figure 2B), before the foramen primum is completely closed by the septum. This occurs between the 5th-6th week of embryonic life.
- Simultaneously, another enfolding appears in the roof of the common atrium to the right of the septum primum called septum secundum(Figure 2C). The inferior end of the septum secundum fuses with the lowermost part of septum primum and the endocardial cushion, causing complete separation of the two atria inferiorly (Figure 2D). Incomplete closure of the foramen secundum by the superior end of septum secundum results in formation of foramen ovale.
- Oxygenated blood coming from the inferior vena cava enters the left atrium and is preferentially directed to the right atrium through the foramen ovale by some primitive right atrial structures. This fetal inter-atrial communication (patent foramen ovale) normally closes after birth due to high left atrial pressure.
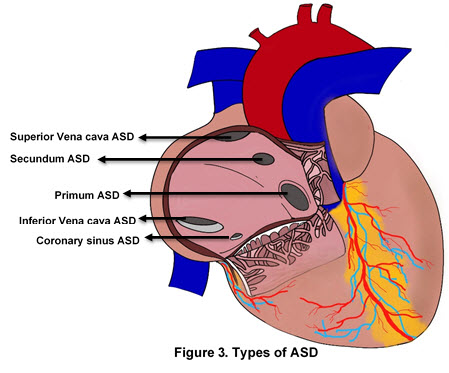
- Defects in the fusion of these structures leads to atrial septal defects (ASD) (Figure 3)
- Primum ASD: Foramen primum remains open
- Incomplete fusion of the septum primum and the atrioventricular endocardial cushions.
- More common in Down syndrome due to disruptions in endocardial cushion development.
- Secundum ASD: Foramen secundum remains open
- Most common type of ASD
- Due to excessive resorption of the septum primum or interrupted septum secundum development.
- Patent foramen ovale: incomplete fusion of the septum secundum and septum primum.
- Most patients are asymptomatic as opening remains functionally closed due to high left atrial pressures.
- Patients with PFO who are in a hypercoagulable state (i.e. pregnancy) are at increased risk for stroke during transient increase in right atrial pressure (i.e. Valsalva, childbirth, constipation). The increase in right atrial pressure may push emboli through the PFO to the systemic circulation.
- ASDs form a persistent shunt between left and right atria while PFOs are covered by a flap that may open and close intermittently.
Ventricular Septation
- The interventricular septum has three components- the muscular, inlet, and infundibular (outlet) septa.
- Muscular interventricular septum forms by proliferation of tissue upward towards the endocardial cushions from the apex of the heart.
- Membranous interventricular septum is formed by the downward growth of the aorticopulmonary septum towards the muscular interventricular septum and posterior-inferior proliferation of tissue from the endocardial cushions.
- Fusion of the membranous and muscular parts of the interventricular septum forms the complete interventricular septum.
- The infundibular septum is in continuation with the atrioventricular canal.
- Subsequent defects in the formation of these different components can lead to muscular, membranous and infundibular ventricular septal defects.
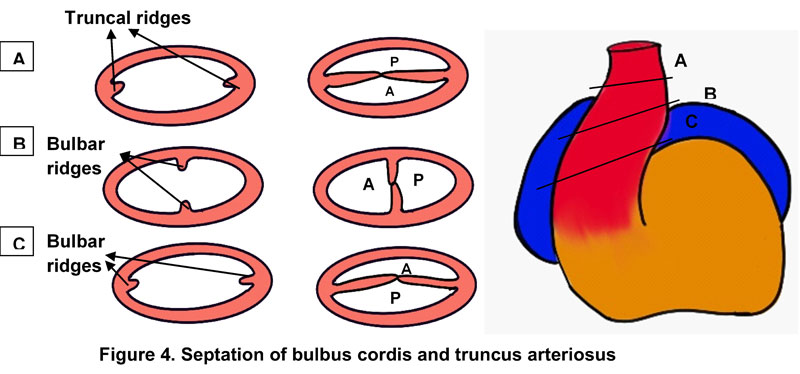
Truncus Arteriosus Septation
- The bulbus cordis elongates and forms three parts- the proximal part forms the trabeculated portion of the right ventricle, the middle part forms conus cordis which is the outflow tract and the truncus arteriosus which forms the ascending aorta and pulmonary trunk.
- Truncus arteriosus undergoes two separate processes: septation and spiraling. There are two swellings along the truncus arteriosus- truncal swellings more distally and conal swellings proximally. The truncal swellings, similar in appearance to endocardial cushions, divide the lumen into the proximal ascending aorta and the pulmonary trunk. Fusion of the truncal and conal swellings establishes the right ventricular origin of the pulmonary trunk and left ventricular origin of the aorta. The aortopulmonary valves develop at the lines of fusion of truncal and conal swellings. (Figure 4)
- Spiralling of the septum allows the pulmonary trunk to cross anterior and to the left of the aorta.
- Failure of neural crest cells to migrate or disruptions to the aorticopulmonary septum leads to congenital heart disease
- Septation defects:
- Persistent truncus arteriosus: Failure of the aorticopulmonary septum to form
- Tetralogy of Fallot: anterior and cephalad deviation of the aorticopulmonary septum
- Double outlet right ventricle
- Spiraling defects:
- d-Transposition of the great vessels