
Molecular Toxicology
Neuropharmacology
-
Director of the Sealy Center for Environmental Health and Medicine (SCEHM)
Professor
Phone: (409) 772-9672
Fax: (409) 772-9642
Email: felaezza@utmb.edu -
1991 – MD, Universita` degli Studi di Torino (Italy)
2001 – PhD, Neuroscience, Emory University, Atlanta, GA -

Our overall goal is to advance the understanding and therapies of complex brain disorders associated with genetic and environmental factors. To this end, we aim to identify key molecular determinants of intrinsic neuronal properties and synaptic integration at the single cell level (Fig. 1) that be mechanistically linked to the biological cause of neuropsychiatric disorders. Our approach includes a pipeline of molecular, cellular and functional modules targeting macromolecular complexes of neuronal ion channels. Our approach spans from in vitro to animal-based assays and employs imaging and electrophysiological methods on a multiscale level.
The primary focus of our research is on voltage-gated Na+ (Nav) channels, a family of nine (Nav1.1-1.9) transmembrane proteins abundantly expressed in the brain. Through a complex network of protein:protein interactions (PPI), the Nav channel complex at the axonal initial segment (AIS) provides the basis for electrical excitability of neurons, enabling transmission, processing and storing of electrochemical signals at single synaptic connections. The Nav channel complex is a vulnerable target of genetic modifications and environmental agents. Mutations targeting the pore-forming α subunit of the Nav channel or its accessory proteins, such as intracellular FGFs, ankyrin-G, ßIV spectrin and neurofascin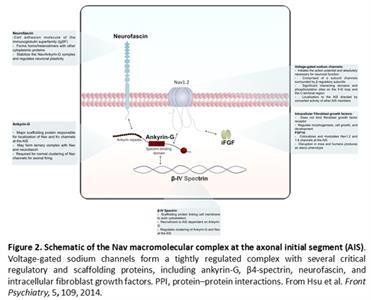 (Fig.
2), are recognized causes or risk factors for epilepsy, mood disorder, autism, depression, schizophrenia, pain and neurodegeneration, making the Nav channel complex one of the most appealing targets for drug development. Yet, the mechanisms underlying
modulation of the Nav channel macromolecular complex in the brain are still poorly understood, limiting the ability to develop molecular interventions against these relevant proteins. Current projects in our laboratory aim at filling knowledge gaps
in this area of research with an emphasis on the intracellular fibroblast growth factors (iFGFs; FGF11-14), a group of versatile and potent regulators of Nav channel biophysics, trafficking and function in the brain.
(Fig.
2), are recognized causes or risk factors for epilepsy, mood disorder, autism, depression, schizophrenia, pain and neurodegeneration, making the Nav channel complex one of the most appealing targets for drug development. Yet, the mechanisms underlying
modulation of the Nav channel macromolecular complex in the brain are still poorly understood, limiting the ability to develop molecular interventions against these relevant proteins. Current projects in our laboratory aim at filling knowledge gaps
in this area of research with an emphasis on the intracellular fibroblast growth factors (iFGFs; FGF11-14), a group of versatile and potent regulators of Nav channel biophysics, trafficking and function in the brain. 
Drug discovery: targeting PPI within the Nav channel complex as a new drug discovery approach. We seek new probes/lead compounds to modulate stability, trafficking and function of the Nav channel complex by engineering short peptides, peptidomimetics, and small molecules that mimic and/or interfere with the PPI of FGF14 and the neuronal Nav channels: Nav1.1, Nav1.2, and Nav1.6 (Fig. 4). Novel promising drug-like molecules targeting the FGF14:Nav1.6 surface are currently under validation in our laboratory. A successful probe and drug discovery campaign against the FGF:Nav channel complex will have broad applicability for the treatment of channelopathies and brain disorders associated with malfunction of the Nav channel complex. Initially funded by the PhRMA foundation and the Institute for Translational Sciences at UTMB, this project is currently being developed in collaboration with Dr. Jia Zhou, a member of the Chemical Biology Program at UTMB.
Intracellular signaling pathways: identifying kinase networks that govern the Nav channel fate within the neuron. The ability of neurons to generate physiologically diverse action potentials is largely determined by expression levels and sub-cellular localization of different Nav channels. Our initial studies indicated an important role for FGF14 in regulating cellular targeting of neuronal Nav channels. Yet, the cellular pathways upstream of this regulatory mechanism controlling maturation, targeting, recycling, and degradation of the Nav channel complex are still poorly characterized. Through a combination of a luminescence-based high-throughput screening (HTS) of kinase inhibitors, co-immunoprecipitation, mass-spectrometry (LC/MS/MS), patch-clamp electrophysiology and quantitative confocal immunofluorescence in native tissue, we discovered a network of signaling pathways that converges on glycogen synthase kinase 3 (GSK3), a critical kinase pathway for brain disorders. Silencing GSK3 reduces the assembly of the FGF14:Nav channel complex, modifies FGF14-dependent regulation of Na+ currents, and induces dissociation and subcellular redistribution of the native FGF14:Nav channel complex in hippocampal neurons with effects on excitability in the hippocampus and nucleus accumbens (NAc); two brain areas associated with neuropsychiatric disorders and neurodegeneration. This research has changed the view of how increased activity of GSK3, common to neuropsychiatric disorders, might translate into disruption of the brain circuit plasticity and maladaptive behaviors. This project is developed in collaboration with Drs. Thomas Green (AAV in vivo gene silencing), Jay Rudra (peptide design), and Cheryl Lichti (proteomics studies) in the Department of Pharmacology & Toxicology at UTMB.
Circuitry and behavior: intervening with selective probes targeting the Nav channel complex to modify brain circuitries and behavior. Maladaptive plasticity of synaptic circuits is a fundamental mechanism driving early phases of complex brain disorders. Through studies in transgenic animal models and, we have discovered changes in synaptic transmission, plasticity, intrinsic excitability and neurogenesis associated with loss of FGF14 function in the hippocampus, cerebellum and NAc. We found that genetic deletion of Fgf14 leads to cortical network disruption, impaired gamma oscillations and reduced working memory; findings that recapitulate essential features associated with schizophrenia and bipolar disease. These new findings corroborated by functional cluster analysis from human schizophrenia transcriptomics identifies FGF14 as part of a network of brain-disease risk genes involved in the cognitive domain. This broad project is currently developed using transgenic animal models and in vivo genetic silencing; animals are assayed in-house with ex vivo brain slice patch-clamp electrophysiology and confocal imaging. Complementary studies including in vivo EEG, behavioral tests and analysis of post-mortem tissues from human transcriptomics analysis have been pursued through intra- and inter-institutional collaborations with Dr. Thomas Green (AAV in vivo gene silencing), Dr. Herbert Meltzer (human transcriptomics, Northwestern University), Dr. Filippo Tempia (animal behavior, Rita Levi-Montalcini Center for Brain Repair, Turin, Italy) and Dr. Benedetto Sacchetti (in vivo electrophysiological recordings, Rita Levi-Montalcini Center for Brain Repair, Turin).
Enabling technologies and quantitative methods in biology. With multimodal data acquisition, biology needs mathematics, computer science, engineering and ultimately information fusion for meaningful data analysis and interpretation. In response to these needs, we have established collaborations with the Department of Mathematics and the Department of Electrical Engineering at the University of Houston to develop specific imaging analysis algorithms for automatic extraction of cellular shapes and analyte pattern expression and distribution in single neurons in culture (for HTS and high-content screening applications) and in the native tissue. This project is developed in collaboration with Drs. Papadakis, Labate and Prasaad.
Relevance of research for NIEHS: pesticides toxicity in the brain. Recent studies identified increased risk in neurodevelopmental disorders (such as attention deficit disorder) with gestational pesticide exposure, including pyrethroids used in agriculture and households as potent stimulants of the insect Nav channel. Due to species cross reactivity, pyrethroids have known effects on mammalian Nav channels expressed in the brain. Yet, the mechanistic link that associates pesticide exposure to increased risk in neuropsychiatric disorders is poorly understood. We have made progress in this novel area of research investigating the pathology of brain disorders at the protein-environment interface. This project is developed under the umbrella of the Center in Environmental Toxicology at UTMB.
-
- Laezza, F, Doherty, JJ & Dingledine, R 1999. Long-term depression in hippocampal interneurons: joint requirement for pre- and postsynaptic events. Science, 285, 1411-4.
- Laezza, F, Gerber, BR, Lou, JY, Kozel, MA, Hartman, H, Craig, AM, Ornitz, DM & Nerbonne, JM 2007a. The FGF14(F145S) mutation disrupts the interaction of FGF14 with voltage-gated Na+ channels and impairs neuronal excitability. J Neurosci, 27, 12033-44.
- Shavkunov, AS, Wildburger, NC, Nenov, MN, James, TF, Buzhdygan, TP, Panova-Elektronova, NI, Green, TA, Veselenak, RL, Bourne, N & Laezza, F # 2013b. The Fibroblast Growth Factor 14:Voltage-gated Sodium Channel Complex Is a New Target of Glycogen Synthase Kinase 3 (GSK3). J Biol Chem, 288, 19370-85.
- Wildburger, NC, Ali, SR, Hsu, WC, Shavkunov, AS, Nenov, MN, Lichti, CF, LeDuc, RD, Mostovenko, E, Panova-Elektronova, NI, Emmett, MR, Nilsson, CL & Laezza, F.# 2015. Quantitative Proteomics Reveals Protein-Protein Interactions with Fibroblast Growth Factor12 as a Component of the Nav1.2 Macromolecular Complex in Mammalian Brain. Mol Cell Proteomics. 2015 Feb 27. pii: mcp.M114.040055. [Epub ahead of print]
- Alshammari TA., Alshammari MA., Nenov MN., Scala F., Laezza F. FGF14 is required for adult neurogenesis of granule neurons in the dentate gyrus. Molecular Neurobiology, 2015
- Hsu, W. J., Scala, F., Nenov, M. N., Wildburger, N. C., Elferink, H., Singh, A. K., Chesson, C. B., Buzhdygan, T., Sohail, M., Shavkunov, A. S., Panova, N. I., Nilsson, C. L., Rudra, J. S., Lichti, C. F. & Laezza, F. CK2 activity is required for the interaction of FGF14 with voltage-gated sodium channels and neuronal excitability. FASEB journal: official publication of the Federation of American Societies for Experimental Biology, doi:10.1096/fj.201500161 (2016)
- Alshammari, T., Alshammari, M., Nenov, M., Hoxha, E., Cambiaghi, M., Marcinno`, A., James, T., Singh, P., Labate, D., Li, J., Meltzer, H., Sacchetti, B., Tempia, F. & Laezza, F. Genetic deletion of fibroblast growth factor 14 recapitulates phenotypic alterations underlying cognitive impairment associated with schizophrenia. Translational Psychiatry May 10;6:e806. doi: 10.1038/tp.2016.66., (2016)
- Hsu J., Wildburger N., Franklin W., Haidacher J., Cortez I., Lichti, C., Dineley K., Taglialatela G., Denner, L., and Laezza F. PPARgamma agonists rescue increased phosphorylation of FGF14 at S226 in the Tg2576 mouse model of Alzheimer’s disease. Exp Neurol. 2017 Sep;295:1-17. doi: 10.1016/j.expneurol.2017.05.005. impact factor: 3.503
- Ali SR, Liu Z., Nenov M., Folorunso, O., Singh A., Scala F., Chen H., James TF, Alshammari M, Panova-Elektronova NI, White MA, Zhou J., Laezza F. Functional Modulation of Voltage-Gated Sodium Channels by a FGF14-Based Peptidomimetic. ACS Chem Neurosci. 2018 Feb 6. doi: 10.1021/acschemneuro.7b00399.
- Scala F., Nenov MN, Crofton EJ, Singh AK, Folorunso O, Zhang Y, Chesson BC, Wildburger NC, James TF, Alshammari MA, Alshammari TK, Elfrink H, Grassi C, Kasper JM, Smith AE, Hommel J, Lichti C, Rudra J, D'Ascenzo M, Green TA, Laezza F. Environmental enrichment and social isolation mediate neuroplasticity of medium spiny neurons through the GSK3 pathway. Cell Reports 2018 Apr 10;23(2):555-567. doi: 10.1016/j.celrep.2018.03.062.