Obstructive Cardiac Lesions
This section deals primarily with ventricular outflow tract obstructive lesions. Congenital mitral and tricuspid valve stenosis are relatively rare in children, thus will be not discussed here.
Outflow tract obstruction leads to an increase in the pressure proximal to the lesion and secondary myocardial hypertrophy. Turbulent flow across the obstruction produces an ejection systolic murmur. Myocardial hypertrophy leads to increased oxygen consumption with decreased exercise tolerance and possible myocardial ischemia and fibrosis. Ventricular dilatation is not a usual response to obstruction, and if present suggests ventricular failure.
Children with mild or moderate obstructive lesions usually have few symptoms. Children with severe obstruction may have low cardiac output symptoms. Newborns with severe obstruction are often PDA dependent to bypass the obstruction.
Arterial switch (PS)
Anatomy
Pulmonary stenosis can be valvular, sub-valvular (infundibular), or supra-valvular.
- The valvular PS is the most common form
- Infundibular PS is usually associated with VSD as in Tetralogy of Fallot.
- Supra-valvular PS is rare and may be associated with congenital rubella or William's syndrome. It may occur as a complication of a previous arterial switch surgery.
- Peripheral pulmonary artery stenosis may result from congenital rubella or associated Alagille or Noonan syndromes
Clinical presentations
Patients with mild or moderate PS are usually asymptomatic. Severe PS may present with decreased cardiac output symptoms such as exertional dyspnea and subsequently symptoms of congestive cardiac failure.
Making the diagnosis
- The physical findings depend on the severity of the obstruction.
- A right ventricular tap and a systolic thrill may be present at the ULSB.
- An ejection systolic murmur is heard best at the ULSB radiating to the back. Wide (and variable) splitting of S2 is commonly present. An ejection systolic click indicates valvular stenosis with post-stenotic dilation of the main pulmonary artery.
- Hepatomegaly may be present if CHF develops.
- The EKG is normal in mild PS. RVH and right axis deviation are usually present in moderate or severe cases. Right atrial enlargement indicates elevated right ventricular filling pressure. Severe PS may cause RV strain pattern (deep inverted T wave and ST segment depression) in right precordial leads. Newborns with critical PS may show LVH because of hypoplastic RV and large LV.
- Chest radiograph usually shows normal heart size with a prominent pulmonary artery segment because of post-stenotic dilatation. Cardiomegaly indicates CHF.
- Echocardiography demonstrates the level and degree of stenosis and the presence of PDA.
Management
Balloon valvuloplasty may be done to relieve the valvular obstruction in moderate or severe cases. Surgery is indicated in supra-valvular and sub-valvular PS or if valvuloplasty is unsuccessful. Infants with critical PS need PGE1 to maintain the ductal patency before intervention.
Aortic Stenosis (AS)
Anatomy
Aortic stenosis may be valvular, supra-valvular or sub-valvular.
- Supra-valvular AS may be associated with Williams syndrome and after arterial switch procedure.
- A discrete fibrous ring, or membrane, may cause sub-valvular obstruction.
- Bicuspid aortic valve is a common cause of AS in patients with Turner Syndrome.
Clinical presentation
Aortic stenosis is more common in males. Children with mild or moderate AS are usually asymptomatic. Severe AS may be accompanied by exertional chest pain, decreased exercise tolerance or episodes of syncope. Infants with critical AS may present with CHF early in life.
Making the diagnosis
- Patients with AS are usually acyanotic unless critical AS is present.
- The pulse pressure may be narrow in severe AS. Patients with supra-valvular AS may have higher BP in the right arm than the left due to preferential streaming (Coanda effect) to the right subclavian artery.
- A systolic thrill may be palpable at the URSB or the supra-sternal notch and radiates to the carotid arteries.
- A systolic ejection murmur is heard at the ULSB and URSB and radiates to the carotid arteries.
- An early diastolic murmur may indicate the development of aortic insufficiency.
- A newborn with critical AS who presents with CHF and poor peripheral pulses may have a faint murmur or no murmur at all.
- "Elfin facies" associated with mental retardation and personality changes suggest William's syndrome and supra-valvular AS.
- EKG is usually normal in mild AS. LVH indicates moderate or severe AS and may be associated with a strain pattern (inverted T waves in left precordial leads).
- Chest radiograph usually shows normal heart size. A prominent aortic knob indicates post-stenotic dilation in valvular AS. Cardiomegaly, if present, indicates severe or critical AS associated with CHF.
- Echocardiography is useful in determining the type and the severity of the lesion.
- Cardiac catheterization is rarely needed for diagnosis.
Management
Mild or moderate AS requires only supportive care. Severe valvular AS can be treated by balloon valvuloplasty. Subvalvular and supravalvular aortic stenosis are not amenable to balloon valvuloplasty and may need surgical intervention.
PGE1 infusion is essential to maintain the ductal patency in neonates with critical AS.
Surgery may involve simple valvotomy or valve replacement with a prosthesis or homograft. Another alternative is the Ross autograft procedure in which the patient's normal pulmonary valve is excised and placed in the aortic position and a homograft replaces the pulmonary valve.
Coarctation of the Aorta
Coarctation of the aorta is usually congenital, but may be acquired as in Takayasu arteritis. It may also be associated with Turner or William's syndromes, and with other CHD such as bicuspid aortic valve, VSD, or double outlet right ventricle.
Anatomy
Congenital coarctation is almost always juxta-ductal (opposite to the ductus arteriosus), and may be either discrete or tubular
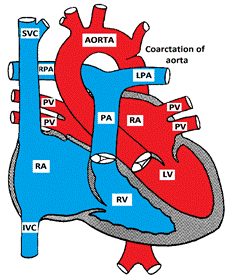
Figure showing Coarctation of the Aorta
Pathophysiology
Coarctation of the aorta causes mechanical obstruction of blood flow from the left ventricle. The pressure proximal to the coarctation is usually elevated. Collateral vessels may develop to bypass the obstruction; the most common collaterals are the internal mammary and the intercostal arteries. LVH develops in response to the elevated pressure proximal to the coarctation.
Clinical presentation
Most patients with coarctation are asymptomatic. Some patients may present with symptoms of either upper extremity hypertension (headaches, blurred vision or frequent nose bleeds) or symptoms of reduced blood flow to the lower extremities (exercise induced claudication). In infancy, severe coarctation may present with CHF as the ductus arteriosus closes. If it is not diagnosed and treated promptly, acidosis, shock and death may occur.
Making the diagnosis
- The hallmarks of coarctation of the aorta are weak, delayed, or absent femoral pulses and a systolic pressure in the lower extremity that is lower than in the upper extremity by >10 mmHg.
- A prominent left ventricular impulse or heave is commonly palpated.
- A systolic ejection click is heard if there is a bicuspid aortic valve.
- A systolic ejection murmur is usually present at the ULSB and the left subscapular region.
- In older children with unrepaired coarctation, continuous murmurs may be heard over the back and chest due to collateral blood flow.
- The EKG varies according to age. RVH is the rule in infancy and LVH develops by the second or third year of life.
- Chest radiograph in neonates with severe coarctation shows severe cardiomegaly and pulmonary edema. Older children may have the "inverted E or 3" sign which is the ascending aortic knob and the post-stenotic dilation. Rib notching from dilated collateral vessels may be present in children more than five years of age.
- Echocardiography is helpful in delineating the lesion. Magnetic resonance angiography is also helpful in making the diagnosis. Cardiac catheterization is rarely needed.
Management
In asymptomatic patients without hypertension, elective surgery or angioplasty is recommended at 18 to 24 months of age. The presence of significant hypertension or CHF in infancy indicates the need for early repair. Neonates with CHF may also need medical management including PGE1 to keep the ductus arteriosus open.
Quick Checks


