Corrected Transposition of the Great Arteries (L-TGA)
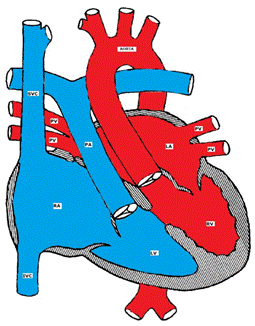
Figure showing Corrected Transposition of the Great Arteries (L-TGA)
Pathology
L-TGA is a rare form of CHD (less than 1%). Both atrio-ventricular and ventricular arterial relationships are reversed, i.e. the right atrium is connected to a morphologic LV that is located on the right and the left atrium is connected to a morphologic RV located on the left (ventricular inversion). The great arteries are also transposed, i.e. the aorta is connected to the morphologic RV on the left and the pulmonary artery is connected to the morphologic LV on the right (Figure). The aortic valve is anterior and to the left of the pulmonary valve. Hemodynamically, both systemic and pulmonary circulations are in series and the patients are usually asymptomatic unless there are other associated defects. L-TGA is commonly associated with other CHD such as VSD (50%), pulmonary stenosis, left AV valve (tricuspid) insufficiency, complete AV block (AVB) and other arrhythmias.
Making the diagnosis
- Patients with L-TGA are usually asymptomatic unless they have other CHD or arrhythmias.
- CHF may develop later as the systemic RV fails to pump against a high systemic vascular resistance.
- Signs and symptoms of associated CHD or arrhythmia may be present.
- On physical examination, the S2 is loud and single (the aorta is anterior to the PA).
- A holosystolic murmur at the LLSB may indicate left AV valve (tricuspid) insufficiency.
- EKG shows absent Q waves in left precordial leads and their presence in V4R (due to reversed septal depolarization), AV block, WPW or other arrhythmias may be present.
- CXR shows a straight left upper cardiac border formed by the ascending aorta.
- Segmental anatomy can be shown by echocardiography. Other associated CHD can also be detected.
Management
Medical: management of CHF and arrhythmias if present. Strenuous sports restriction is indicated to prevent strain of the systemic right ventricle.
Surgical: repair of associated CHD (VSD, PS, tricuspid valve replacement). Pacemaker implantation may be needed for AVB. Double switch procedure (arterial and atrial) has been tried to treat the failing RV (Figures). Heart transplantation may be considered in some cas
Persistent Truncus Arteriosus (TA)
Truncus arteriosus is sometimes associated with DiGeorge syndrome (deletion of chromosome 22q.11) which is characterized by hypocalcemia and reduced T-lymphocyte function. Extra-cardiac abnormalities are present in 25-40% of patients.
Anatomy
A single large trunk overrides a large VSD and gives rise to the coronary arteries, pulmonary arteries, and the brachiocephalic arteries (Figure).
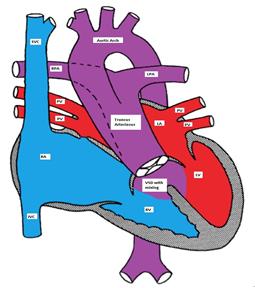
Truncus Arteriosus, with the main pulmonary artery arising from the common trunk
Pathophysiology
TA has a single ventricle physiology as the VSD is usually large (one ventricle pumps blood to both systemic and pulmonary circulations with complete mixing of blue and red blood). The truncal valve is frequently abnormal. In the neonatal period, the pulmonary vascular resistance is elevated and the pulmonary blood flow may be limited. This may cause mild and transient cyanosis due to a right-to-left shunt. As the pulmonary vascular resistance drops, the pulmonary blood flow significantly increases causing CHF symptoms.
Clinical Presentation
Physical Examination: mild cyanosis may be detected along with a wide pulse pressure and cardiomegaly. A single S2 (one semilunar valve) and a systolic ejection murmur due to increased flow through the truncal valve may be heard. An apical diastolic murmur may be heard due to increased flow through the mitral valve (relative mitral stenosis). An early diastolic murmur may indicate truncal valve insufficiency.
Making the Diagnosis
- EKG may show biventricular hypertrophy.
- CXR shows increased pulmonary vascular markings and prominent ascending aorta. It may also be suggestive of right aortic arch (25% of cases).
- Echo is diagnostic and shows associated cardiac anomalies.
Management
Medical management of CHF may be tried until surgical repair can be performed.
Surgical repair may include:
- Early surgical correction is preferred. The VSD is closed so that the LV is connected to the truncus and the pulmonary artery is detached from the truncus and is connected to the right ventricle via a conduit.
- If early surgical repair cannot be done, banding of the pulmonary artery is performed to limit the blood flow to the lung and to control CHF.
Tetralogy of Fallot (TOF)
Tetralogy of Fallot is the most common cyanotic CHD (over 50% of all cases of cyanotic CHD; ~ 10% of all cases of CHD).
Anatomy (figure)
TOF has four abnormalities (hence the term 'tetralogy):
- Ventricular septal defect (mal-alignment)
- Over-riding aorta
- Right ventricular outflow tract obstruction (RVOTO)
- Right ventricular hypertrophy (RVH)
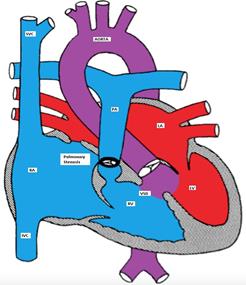
Figure showing tetralogy of Fallot (TOF) with large mal-aligned VSD, overriding aorta,
right ventricular outflow tract obstruction and right ventricular hypertrophy
Pathophysiology
The VSD is nonrestrictive (both ventricles have equal pressures). The direction of the blood flow across the VSD depends on the relative systemic and right ventricular outflow tract (RVOT) resistances.
Clinical manifestations
TOF patients usually present with a heart murmur at birth with variable degree of cyanosis (depending on the degree of RVOTO). Many patients are completely acyanotic (pink TOF). There is a right ventricular tap at the left sternal border, systolic ejection murmur due to RVOTO, and single S2. The VSD is usually non-restrictive and does not produce a heart murmur. Clubbing of digits may be seen in untreated TOF with prolonged cyanosis.
Making the Diagnosis
More Information
TOF with Pulmonary Atresia
Pulmonary atresia may occur in 15% of patients with TOF and may be associated with hypoplasia of the peripheral pulmonary arteries. In this lesion, the pulmonary blood flow depends on the patency of the ductus arteriosus. Collaterals develop between the systemic and pulmonary circulations. The patients are usually cyanotic at birth and the degree of cyanosis depends on the degree of mixing of both circuits. The patency of the ductus arteriosus has to be maintained by PGE-1 until surgery is performed.
TOF with absent pulmonary valve
This condition occurs in about 2% of patients with TOF. The pulmonary valve is usually dysplastic, regurgitant and stenotic. There is aneurysmal dilation of the proximal pulmonary arteries, which may compress the major airways causing obstruction, especially in supine position.
Clinically, the patients have respiratory distress in supine position and mild cyanosis which may disappear if CHF develops due to decrease in the pulmonary vascular resistance. To-and-fro murmur due to PS and PI is classic. Surgery is usually needed to reduce the airway compression and to repair the TOF
- EKG: RAD, RVH
- CXR: 'Boot shaped' heart due to concave main pulmonary artery segment and upturned RV apex due to RVH (Figure)
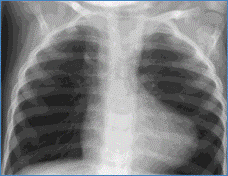
Chest X-ray showing boot shaped heart in tetralogy of Fallot
- Echocardiography is diagnostic. 25% of patients have right aortic arch and 5% have coronary artery anomalies.
Tet spells (hypercyanotic or hypoxic spells): (hence the term 'blue baby)
Tet spells are characterized by paroxysmal cyanosis, tachycardia, tachypnea and irritability. These spells are caused by reversal of the shunt across the VSD so the blood flows from the right to the left ventricle due to an increase in the RVOTO resistance. Initially, the systolic murmur gets louder and then diminishes as the RVOTO progresses to near complete obstruction. Tet spells are more common in the warm morning hours when the systemic vascular resistance is low due to increased parasympathetic tone and decreased intravascular volume; a warm bath can have a similar effect. The RVOTO is worsened by increased catecholamine secretion due to irritability, hunger, or wet diapers.
Hypercyanotic spells need urgent treatment because if left untreated, metabolic acidosis, seizures or cerebrovascular accidents or even death may occur. The concept of treatment is to reverse the mechanism of the hypoxic spells. It is important to remove any source of irritation for the infant to decrease the catecholamine surge and worsening of the RVOTO.
While the mother is comforting the baby (nursing, bottle feeding), the baby is placed in a knee-to-chest position to increase systemic vascular resistance. Blow-by O2 is provided to enhance the oxygenation of blood and increase the systemic vascular resistance. If the spell continues, morphine sulphate (0.2 mg/kg) may be given intramuscularly to calm the patient and relax the obstruction of the RVOT. An IV line should be inserted and isotonic fluid boluses should be administered and may be repeated until the heart rate normalizes.
Acidosis should be treated with sodium bicarbonate. If the spell continues, vasoconstrictors such as phenylephrine may be tried. Intravenous beta blockers (propranolol or esmolol) may be used to decrease the cardiac contractility and hence the RVOTO. Beta blockers also slow down the heart rate and therefore enhance the coronary perfusion. If the spell continues despite all these measures, endotracheal intubation and general anesthesia are indicated in preparation for urgent surgical intervention.
Management
If the patient is pink and has never had a cyanotic spell, elective surgery is usually performed in the first few months of life by closing the VSD and relieving the RVOTO. If the patient is cyanotic or develops a cyanotic spell, intervention is necessary in the neonatal period. If neonatal repair cannot be done because of prematurity or an unstable medical condition, palliative Blalock-Taussig shunt (anastomosis between the subclavian artery and pulmonary artery)(Figure) or its modification, a shunt between the innominate and pulmonary arteries, is performed to bypass the RVOTO.
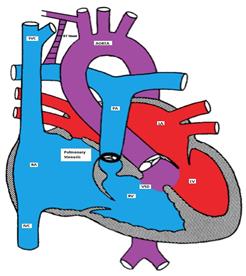
Figure showing tetralogy of Fallot (TOF) with a BT shunt between the right subclavian and the right pulmonary arteries
TOF with Pulmonary Atresia:
Pulmonary atresia may occur in 15% of patients with TOF and may be associated with hypoplasia of the peripheral pulmonary arteries. In this lesion, the pulmonary blood flow depends on the patency of the ductus arteriosus. Collaterals develop between the systemic and pulmonary circulations. The patients are usually cyanotic at birth and the degree of cyanosis depends on the degree of mixing of both circuits. The patency of the ductus arteriosus has to be maintained by PGE-1 until surgery is performed.
TOF with Absent Pulmonary Valve:
This condition occurs in about 2% of patients with TOF. The pulmonary valve is usually dysplastic, regurgitant and stenotic. There is aneurysmal dilation of the proximal pulmonary arteries, which may compress the major airways causing obstruction, especially in the supine position.
Clinically, the patients have respiratory distress in the supine position and mild cyanosis which may disappear if CHF develops due to a decrease in the pulmonary vascular resistance. To-and-fro murmur due to PS and PI is classic. Surgery is usually needed to reduce the airway compression and to repair the TOF.
Total Anomalous Pulmonary Venous Connection (TAPVC)
TAPVC accounts for about 1% of all CHD. It is also referred to as total anomalous pulmonary venous return (TAPVR) which is less accurate as the connection may be obstructed and there may be diminished or no pulmonary venous return.
In TAPVC, the pulmonary veins connect to the right atrium or great veins (SVS or IVC) instead of connecting to the left atrium. An atrial communication is necessary to maintain life.
Pathophysiology
In the right atrium, both oxygenated and deoxygenated blood completely mix, and the mixture is then distributed to other cardiac chambers. The oxygen saturation is characteristically the same in all cardiac chambers. There are four different types of TVAPC:
- Supracardiac: The pulmonary veins connect via a vertical vein to the innominate vein and the SVC (Figure).
- Cardiac: The pulmonary veins connect to coronary sinus or the right atrium (Figure).
- Infracardiac: A common pulmonary vein crosses the diaphragm and connects to the portal vein, ductus venosus, hepatic vein or the IVC. (Figure).
- Mixed type is a combination of the above.
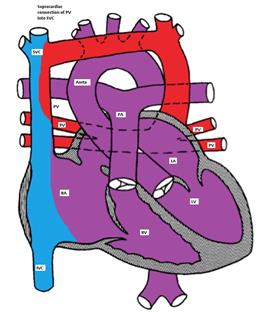
Figure showing supracardiac type TAPVC with drainage of pulmonary veins into superior vena cava (SVC) via vertical vein, which connects to the innominate vein and finally drains into SVC
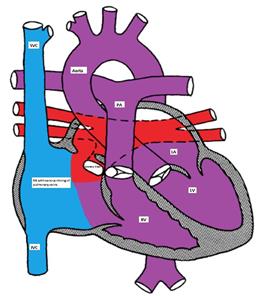
Figure showing the cardiac type of TAPVC with drainage of pulmonary veins directly into right atrium via coronary sinus
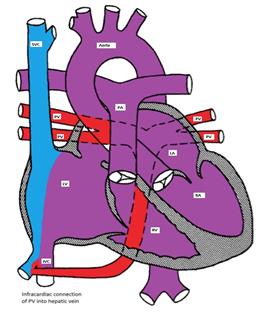
Figure showing infracardiac type TAPVC with drainage of pulmonary veins into inferior vena cava via hepatic vein
Clinical manifestations
The clinical manifestations depend on the presence or absence of obstruction to the pulmonary venous connection. If there is no obstruction, mild cyanosis, CHF and frequent pulmonary infection are the common manifestations. With obstruction, there is marked cyanosis, respiratory distress and pulmonary congestion.
Making the Diagnosis
- CXR shows pulmonary edema.
- Cardiomegaly with a "snowman" sign may be seen in supracardiac type (Figure).
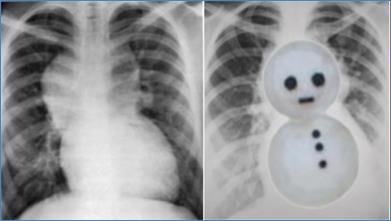
Chest X-ray showing snowman sign of supracardiac TAPVC
Management
Medical management may be tried in patients with TAPVC without obstruction in the form of diuretics and correction of metabolic acidosis. PGE1 causes pulmonary vasodilatation and should be avoided as it may worsen the CHF. All patients with TAPVC will need surgical repair as soon as the diagnosis is made where the pulmonary veins are re-implanted in the left atrium.


